Selecting the right phosphorus-based agent requires a clear-eyed assessment of phase-state trade-offs. While both materials serve as critical reducing agents and flame retardant precursors, their efficacy is dictated by the thermal and chemical environment of the application.
Liquid Dynamics: The Role of Hypophosphorous Acid
In aqueous systems, the performance of the reducing agent depends entirely on concentration stability and the absence of metallic catalysts. For procurement teams vetting Hypophosphorous Acid suppliers, the primary concern is the “assay drift.”
Liquid is volatile
The acid is a powerful monobasic acid. In electroless nickel plating, it acts as the primary electron donor to reduce metal ions into a uniform coating. However, if the supplier does not control for trace iron or heavy metals, the acid begins to disproportionate. This leads to the formation of phosphine gas and phosphoric acid—effectively killing the bath’s efficacy and creating a significant safety hazard.
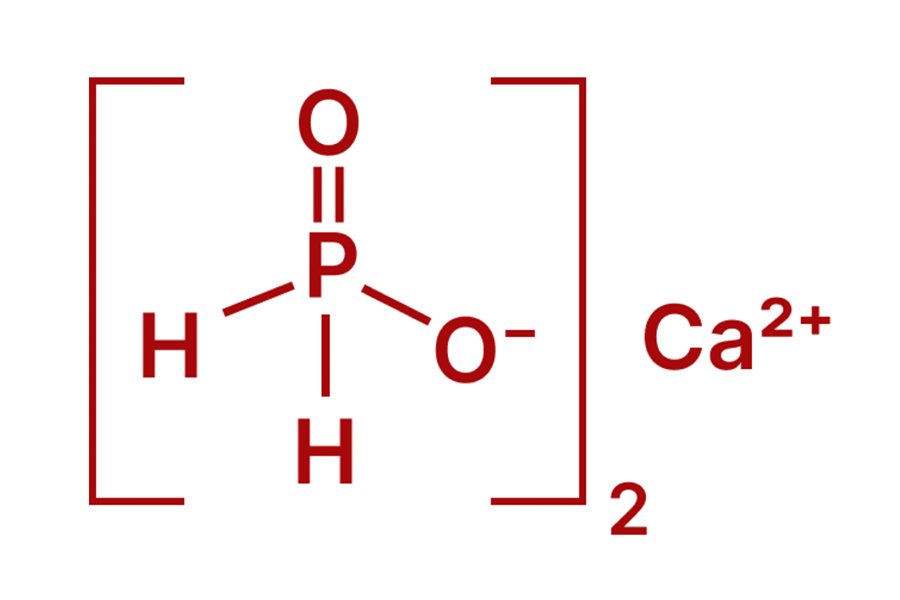
Solid-State Stability: Calcium Hypophosphite
When the application moves into the realm of engineering plastics or cement additives, the liquid acid is no longer viable. Calcium Hypophosphite becomes the standard. It provides the same hypophosphite ion but in a thermally stable, white crystalline powder.
Solids prioritize structural integrity
For flame retardancy, this salt is often integrated into polyamides. Unlike the liquid acid, it does not introduce water into the polymer melt. Water is the enemy of high-temperature extrusion; it causes “splay” and mechanical degradation. By using a solid salt, manufacturers can achieve a UL 94 V-0 rating without compromising the tensile strength of the finished part.
Comparative Efficacy Benchmarks
The choice between these two agents is a tactical trade-off between “reactivity” and “stability.”
Reaction Velocity:
The liquid acid is more reactive and works at lower temperatures, making it ideal for pharmaceutical synthesis and fine chemical intermediates.
Thermal Window:
The calcium salt can withstand temperatures up to 300°C before significant decomposition begins. It is the “heavy lifter” for high-heat manufacturing.
Byproduct Profile:
The acid leaves behind a liquid acidic residue that may require neutralization. The calcium salt leaves behind a solid calcium phosphate char, which actually helps reinforce the fire-resistant barrier in plastics.
The Vendor Audit: Purity is Performance
Why do some batches fail while others thrive?
It usually traces back to the “insoluble” content in the solid salt or the “color value” of the liquid acid. A low-tier calcium salt might contain residual calcium sulfate. These particles act as stress-concentrators in a plastic part, causing it to snap under pressure. Similarly, a yellowish tint in hypophosphorous acid indicates oxidation—a sign that the reducing power of the batch is already depleted.
Final Technical Positioning
Do not treat these chemicals as interchangeable commodities.
If your process involves moisture-sensitive organic reactions, the solid calcium salt—properly dried—is your safeguard. If you are running a high-volume plating line where instant solubility and high electron density are required, the liquid acid is the only logical choice. Audit your Hypophosphorous Acid suppliers for their heavy metal limits and verify the particle size of your calcium salts.
Precision over price
In the 2026 industrial landscape, the cost of a “bad batch” far exceeds the margin gained by sourcing subpar purity levels. State your analytical requirements upfront; ensure the chemistry matches the heat of your process.